 /Charles_and_Gay-Lussacs_Law-56a129b25f9b58b7d0bca3df.gif) 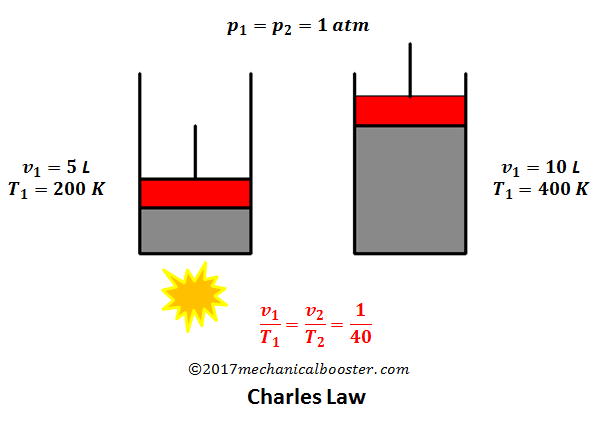
At an altitude of 1 km (90 kPa), assuming the patient remains at a constant temperature, we can rearrange the formula to V2= (P1 They have a pneumothorax with a volume of 1500 mL at sea level (101.3 kPa). For example, a patient with a simple pneumothorax being airlifted to their local hospital. Using Boyle’s law, P1V1 = P2V2, we can calculate the change in volume at different altitudes. When ascending from depth, if a diver holds their breath, the gases in their lungs will expand and can cause barotrauma, arterial gas embolism, mediastinal emphysema, or even pneumothorax. 8200 feet).īoyles law also explains the use of saline in the cuff of an endotracheal tube during hyperbaric therapy to prevent an air leak due to the reduction of volume as pressure increases. It is estimated that an expansion of up to 30% for a closed volume of gas in the human body, e.g., a bulla, can be expected after ascending from sea level to an altitude of 2.5 km (approx. 5000 feet) from sea level, an effect which may prompt thoracostomy before helicopter transfer to prevent transition to a tension pneumothorax. In one artificial pneumothorax model, a 40 mL pneumothorax increased in volume by up to 16% at 1.5 km (approx. This effect can be demonstrated by observing the expansion of a sealed bag of potato chips on an ascending commercial flight. As altitude increases, ambient pressure decreases, and therefore, by Boyle’s Law, volume expansion occurs in enclosed spaces. Assuming the pressure remains constant throughout, find the new volume of the balloon.Boyle’s law can be used to describe the effects of altitude on gases in closed cavities within the body, and to calculate the total intra-thoracic gas volume by body plethysmography. The balloon is then heated to a temperature of 65 ˚C. Problem 2: A balloon is filled to a volume of 3.2 L at a temperature of 25 ˚C. Problem 1: What change in volume results if 4 L of oxygen is cooled by 6.0 ☌ from 120 ☌? When brought back inside a warm room, the balloon gets back to its original shape. It crumbles when the balloon is taken out of a house on a cold day. A helium balloon behaves similarly to a tire.The high temperature outside increases the air temperature inside. On a cold day, the low temperature outside reduces the air temperature inside. The volume of air inside a tire is affected by the outside temperature.The air expands, and its pressure forces repair the dent. The high temperature of water raises the air temperature inside the ball. 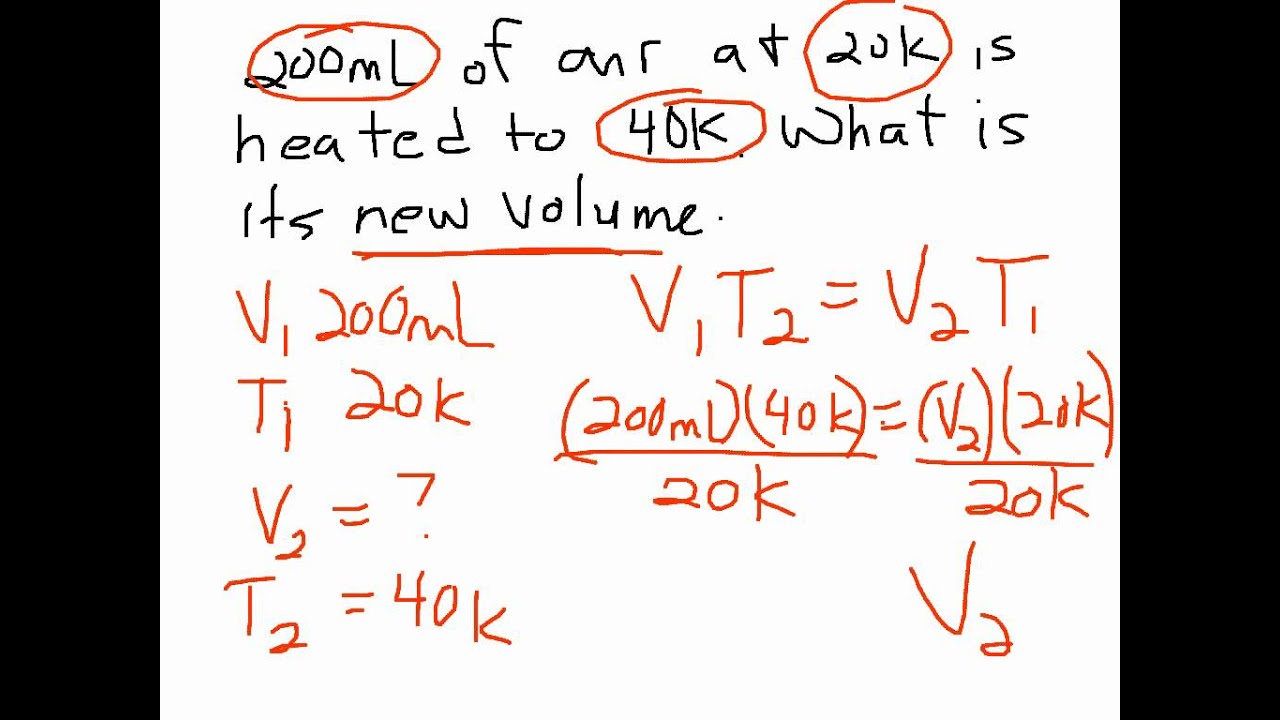
The dent in a ping pong ball can be repaired by immersing it in warm water.The air inside the tube is heated, and its temperature rises. The opposite happens on a hot summer day. Hence, the air temperature inside the tube is low, and the tube shrinks. On a cold winter day, the water temperature is near freezing. Hence, running and jogging become challenging to do in winter. In winter, due to cold air inside them, the lungs shrink. The envelope of air inside the balloon is lighter than the air outside, making it easier for the balloon to rise. 
The air expands, thereby increasing the volume and decreasing the density.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
